Multiplex Reactions
To save time, money, and materials, much work has gone into developing procedures whereby test DNA can be "incubated" with a combination of PCR primers for several different markers at one time. Primers are designed to bind at only one spot in the genome so that during the PCR each primer can be busily dissecting out and amplifying its own STR marker exclusively.
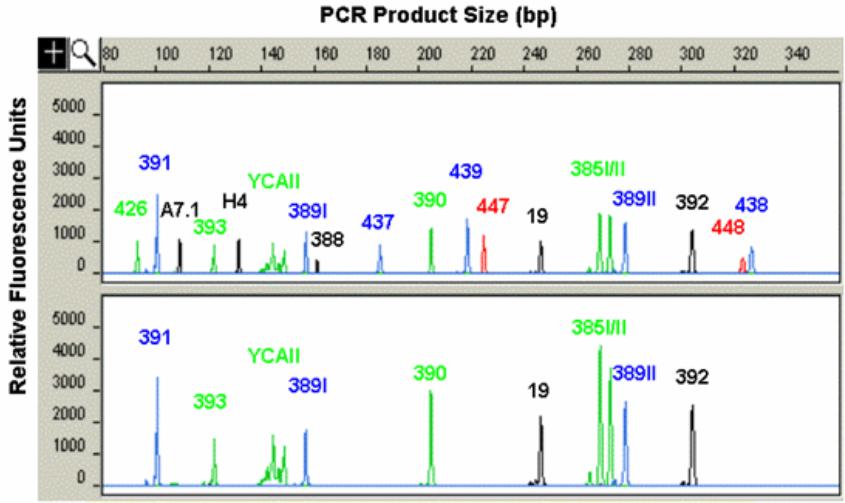
The task of sorting out the results at the end is facilitated by adding various colored fluorescent tags to the fragments. Fluorescent labeling of DNA fragments may be performed in several ways. The most common method is to incorporate a fluorescent dye on the 5'-end of a PCR primer so that during PCR amplification either the forward or the reverse strand of DNA will be labeled.
At the end of the PCR, the mixture will be run through a gel electrophoresis slab or an array of microtubules each filled with a gel. Since each amplified fragment has a discrete length that depends on the number of repeat motifs, it will move through the gel at a different rate. The colors of the fluorescent labels are coordinated with the size ranges of the markers so that several markers in the same size range can be run together, each labeled with a different color.